Tagged: health
Mystery at Sea: The Deadly Hantavirus Outbreak on an Expedition Cruise

A rare and terrifying medical emergency has been unfolding over the last few weeks on an expedition cruise ship. What started as an adventure to some of the world’s most remote locations quickly turned into a deadly crisis involving a little-known, yet incredibly dangerous, pathogen: the Hantavirus.
The Unfolding Crisis on the MV Hondius On April 1, 2026, the Dutch-flagged MV Hondius, operated by Oceanwide Expeditions, departed from Ushuaia, Argentina, with 147 passengers and crew on board. The vessel was scheduled to tour the ecologically diverse and remote regions of Antarctica and the South Atlantic islands.
Just days into the voyage on April 6, a 70-year-old Dutch passenger fell ill with a fever, headache, and mild diarrhea. His condition rapidly deteriorated into respiratory distress, and he tragically died on board on April 11. Over the following weeks, a terrifying cluster of severe respiratory illness emerged among the passengers. His 69-year-old wife, who had disembarked with his body at St. Helena, died shortly after arriving at an emergency department in South Africa. Another British passenger fell critically ill with pneumonia and had to be medically evacuated to an ICU in South Africa, while a fourth passenger, a German woman, died on the ship on May 2.
It took nearly a month from the first illness for laboratory officials in South Africa to finally confirm the culprit: Hantavirus. In total, the outbreak has resulted in three deaths, one critical hospitalization, and three other suspected cases remaining on the ship.
Currently, the ship is moored off the coast of Cape Verde waiting for emergency medical evacuations, while passengers remain under maximum physical distancing lockdowns in their cabins. Authorities suspect the initial exposure may have occurred when the first two victims traveled through Argentina before boarding, as the “Andes” strain of the hantavirus is endemic to South America.
Summary of cases from World Health Organization (WHO):
Case 1: An adult male developed symptoms of fever, headache, and mild diarrhoea on 6 April 2026 while on board the ship. By 11 April, the case developed respiratory distress and died on board on the same day. No microbiological tests were performed. The body of the passenger was removed from the vessel to Saint Helena (a British Overseas Territory) on 24 April.
Case 2: An adult female, who was a close contact of case 1, went ashore at Saint Helena on 24 April 2026 with gastrointestinal symptoms. She subsequently deteriorated during a flight to Johannesburg, South Africa, on 25 April. She later died upon arrival at the emergency department on 26 April. On 4 May, the case was subsequently confirmed by PCR with hantavirus infection. Contact tracing for passengers on the flight has been initiated.
Cases 1 and 2, had travelled in South America, including Argentina, before they boarded the cruise ship on 1 April 2026.
Case 3: An adult male presented to the ship’s doctor on 24 April 2026 with febrile illness, shortness of breath and signs of pneumonia. On 26 April, his condition worsened. He was medically evacuated from Ascension to South Africa on 27 April, where he is currently hospitalised in an Intensive Care Unit (ICU). Laboratory testing on an extensive respiratory pathogen panel was negative; however, polymerase chain reaction (PCR) testing confirmed hantavirus infection on 2 May 2026. Serology, sequencing and metagenomics are ongoing.
Case 4: An adult female, with presentation of pneumonia, died on 2 May 2026. The onset of symptoms was on 28 April, with fever and a general feeling of being unwell.Three suspected cases have reported high fever and/or gastrointestinal symptoms and remain on board. Medical teams in Cabo Verde are evaluating the patients and collecting additional specimens for testing.
What Exactly is Hantavirus? Hantaviruses are a family of viruses that are spread mainly by rodents. People typically contract the virus by coming into close contact with the urine, feces, or saliva of infected rodents, such as rats and mice. While rare, the virus can also be transmitted through rodent bites or scratches.
Generally, hantaviruses do not spread from person to person. However, there have been rare but documented instances of limited human-to-human transmission associated specifically with the Andes virus found in South America. However, there is a rare exception. In these uncommon instances, the virus can spread between people through close and prolonged contact in community settings. There have also been rare instances of secondary infections documented among healthcare workers in healthcare facilities.
Symptoms to Watch Out For Depending on the specific virus and region, hantaviruses typically cause two severe syndromes:
- Hantavirus Pulmonary Syndrome (HPS): Found mostly in the Western Hemisphere (including the U.S., Canada, and South America), this deadly disease severely affects the lungs. Early symptoms appear 1 to 8 weeks after exposure and look a lot like the flu—fatigue, fever, and severe muscle aches, particularly in the thighs, hips, and back. After 4 to 10 days, the late-stage symptoms set in, and patients develop coughing and shortness of breath as their lungs rapidly fill with fluid. HPS is extremely dangerous, with a fatality rate of up to 38% to 50% in the Americas.
- Hemorrhagic Fever with Renal Syndrome (HFRS): Found primarily in Europe and Asia, this syndrome severely impacts the kidneys. Symptoms appear within 1 to 2 weeks and include sudden intense headaches, back and abdominal pain, fever, nausea, and blurred vision. As the disease progresses, patients may experience low blood pressure, internal bleeding, acute shock, and acute kidney failure.
Treatment and Prevention Currently, there is no specific antiviral treatment, cure, or vaccine for hantavirus infections. Patients must rely on immediate supportive medical care in an intensive care unit. This care may involve intubation for breathing support in HPS cases, or dialysis to remove toxins from the blood in HFRS cases when the kidneys are failing. Antibiotics are also not routinely used unless a concurrent bacterial infection is suspected. While the antiviral drug ribavirin has shown some efficacy in treating Hemorrhagic Fever with Renal Syndrome (HFRS), it is not effective or licensed for treating the lung-affecting Hantavirus Pulmonary Syndrome.
Because there is no cure, treatment relies entirely on immediate supportive medical care, and early referral to an intensive care unit (ICU) is critical for improving a patient’s chances of survival. This supportive care may involve:
- Rest, hydration, and medications to manage fever and pain.
- Careful fluid administration and blood pressure (hemodynamic) monitoring.
- Respiratory support for patients experiencing breathing difficulties, which can include intubation, mechanical ventilation, or even extracorporeal mechanical oxygenation in severe cases.
- Dialysis for patients experiencing acute kidney failure to help remove toxins from the blood and balance fluids.
To protect yourself from hantavirus, health authorities recommend eliminating or minimizing contact with rodents in your home, workplace, or campsite. This includes sealing holes and gaps to keep rodents out, placing traps to decrease infestations, and immediately cleaning up any easily accessible food that might attract mice and rats. If you suspect you have been exposed to rodents and develop symptoms like fever, deep muscle aches, and severe fatigue, you should see a physician immediately and mention the potential exposure.
The 200x Factor: Why the Newest Veterinary Sedative is Transforming the Drug Crisis

Savage Sisters’ community outreach storefront in the Kensington neighborhood of Philadelphia. (npr.org)
The Invisible Shift: An Introduction
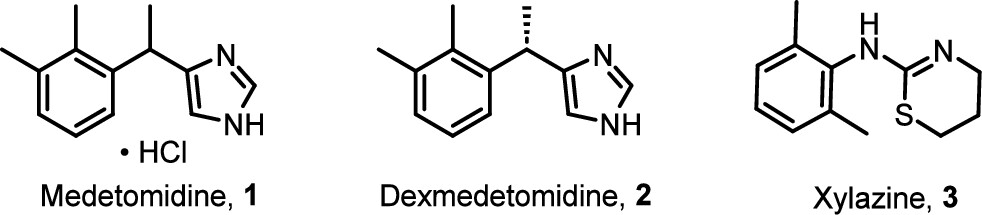
In May 2024, the illicit drug supply in Philadelphia and Chicago underwent a tectonic shift that caught public health systems largely off-guard. For years, the narrative of “Tranq” revolved around xylazine—a veterinary sedative known for leaving users with horrific, necrotic skin ulcers. But a new, stealthier adulterant has arrived: Medetomidine. This isn’t just another additive; it is a pharmacological escalation that mirrors the “China White” (alpha-methylfentanyl) epidemic of the 1970s. Just as China White blindsided the system as a “stealth” fentanyl analog, medetomidine has infiltrated the market under the radar, rendering previous harm-reduction strategies insufficient overnight. This discovery is a game-changer because it signifies a pivot in the supply—moving away from the visible rot of xylazine toward an “invisible” and far more potent cardiovascular killer.
Takeaway 1: The “200x” Potency Leap
Medetomidine represents a massive leap in strength. According to clinical reports, it is between 100 and 200 times more potent than xylazine. While the public is only beginning to understand the dangers of “Tranq,” the illicit market has already evolved into something far more lethal. This extreme potency increases the risk of severe bradycardia (critically slow heart rate), profound hypotension, and respiratory depression. For those on the front lines of harm reduction, this jump in potency means that the “monitoring windows” and supportive care protocols designed for xylazine are now fundamentally inadequate.
“Medetomidine is 100-200 times more potent than xylazine and can cause longer-lasting sedation, low heart rates, and more severe withdrawal symptoms. It is not an opioid but is found in the dope (street opioid) supply.” — Substance Use Philly Report
Takeaway 2: The Bradycardia “Signature”
In recent clusters across Chicago and New Jersey, clinicians have documented a distinct “atypical toxidrome.” Unlike standard opioid overdoses, which primarily shut down the respiratory system, medetomidine leaves a unique cardiovascular footprint. Patients are presenting with heart rates as low as 30–40 beats per minute (bpm)—a level of bradycardia rarely seen with other adulterants. Paradoxically, this is often paired with “hypertensive urgency”—spikes in blood pressure that create a confusing clinical picture for responders used to the “low and slow” profile of heroin.
Clinical Vitals from the ToxIC NOSE Report:
- Heart Rate Range: 34–133 bpm
- Blood Pressure Range: 64/37–170/100 mmHg
Takeaway 3: Why Naloxone Isn’t Enough
The most harrowing aspect for first responders is the “partial response.” Medetomidine is an alpha-2 adrenergic receptor agonist, not an opioid. It induces a deep, heavy sedation by inhibiting noradrenergic neurons in the locus ceruleus—essentially mirroring a state of natural sleep (Stage 2 NREM). This is fundamentally different from the “Mu-receptor” depression of opioids or the “GABAergic” shutdown of benzodiazepines.
When a responder administers naloxone, they may successfully reverse the fentanyl, bringing the patient’s respiratory rate back to normal. However, the patient often remains a “stone-cold” unconscious statue—breathing, but completely unreachable and bradycardic due to the medetomidine. This creates a terrifying clinical limbo where the patient is “saved” from respiratory arrest but remains deeply sedated and cardiac-compromised.
“Because all specimens and samples in this investigation that contained medetomidine also contained natural or synthetic opioids, administering naloxone for all suspected opioid-involved overdoses remains crucial… the effects of medetomidine cannot be reversed with naloxone.” — CDC MMWR Report
Takeaway 4: The “Forbidden” Antidote (Atipamezole)
In the veterinary world, a “cure” for this sedation exists: Atipamezole (Antisedan). It is a highly effective reversal agent used to wake up a bizarrely diverse roster of patients, including okapi, giraffes, red-tailed hawks, koi carp, alligators, and red-eared sliders.
However, Atipamezole is a “forbidden” antidote for humans, presenting a lethal pharmacological catch-22. While it can wake a dog in minutes, administering it to a human who has medetomidine in their system can trigger immediate cardiovascular collapse. This occurs because the drug can cause a sudden drop in blood pressure and profound hypotension while the body is still struggling with reflex bradycardia. We have a way to “wake up” the patient, but the antidote itself could kill them before they open their eyes.
Takeaway 5: The Stealth Takeover of the Supply
The speed at which medetomidine has supplanted xylazine is a testament to the volatility of the illicit market. In Philadelphia, the transition has been near-total in a matter of months. This shift marks a transition from “visible necrosis” (the skin rot associated with xylazine) to “invisible cardiac arrest.” Suppliers are opting for medetomidine likely because its “heavy” sedation profile mimics a more powerful opioid “nod,” even as it silently stresses the user’s heart.
Philadelphia Market Shift (May–November 2024):
- Medetomidine Prevalence: Skyrocketed from 29% to 87%
- Xylazine Prevalence: Plummeted from 97% to 42%
Takeaway 6: The Bizarre Side Effects in Research
Because medetomidine targets the brain’s noradrenergic system so specifically, research has revealed unsettling side effects. In human studies, low doses can actually improve alertness, but the high, uncontrolled doses found on the street likely cause norepinephrine overactivity. This leads to a total collapse of cognitive function; while a user might maintain “selective attention” on a single task, their ability to multitask or respond to their environment vanishes. Furthermore, pharmacological research in monkeys has documented a significant increase in sexual activity following the administration of alpha-2 antagonists, highlighting how deep and unpredictable the neurological impact of these substances truly is.
Conclusion: A New Era of Adulterants
The arrival of medetomidine signals that we have entered a new, more clinical phase of the drug crisis. We are no longer just dealing with opioids; we are managing a supply contaminated with high-potency veterinary anesthetics that require specialized cardiovascular intervention. To survive this, we need a multisector surveillance net—clinicians, toxicology labs, and public health strategists working in a rapid-response loop.
The “Tranq” wounds of yesterday were a warning we could see. The cardiac failures of today are a warning we cannot. As the veterinary-to-street pipeline becomes a permanent fixture of the illicit market, we must ask: Are we prepared for a future where the drug supply evolves faster than our ability to regulate or treat it?
Further Resources